The Impact of GLP-1 on Fasting for Kids with Type 1 Diabetes
Glucagon-like peptide-1 (GLP-1) has been extensively studied in the context of type 2 diabetes, but its role in children with type 1 diabetes (T1D) is not well-defined, and evidence is scarce. A systematic review aimed to compare fasting and postprandial incretin levels in children and adolescents with T1D versus healthy controls and assess their relationship with glycemic outcomes and disease duration in T1D.
What is GLP-1 and its Role in Diabetes Management?
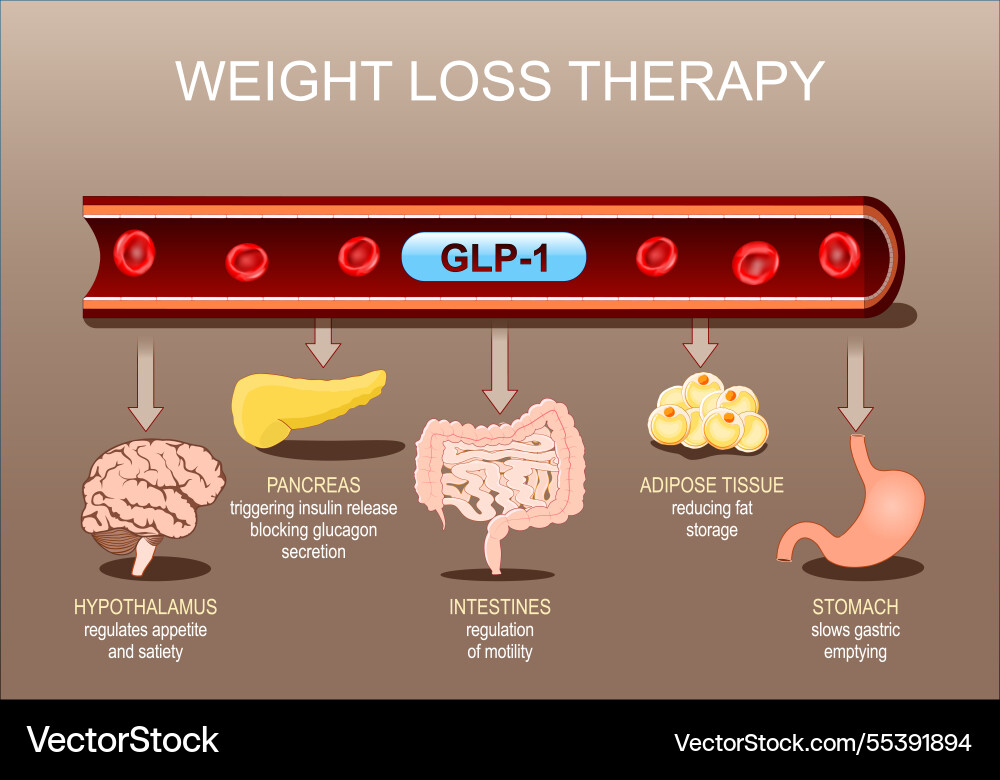
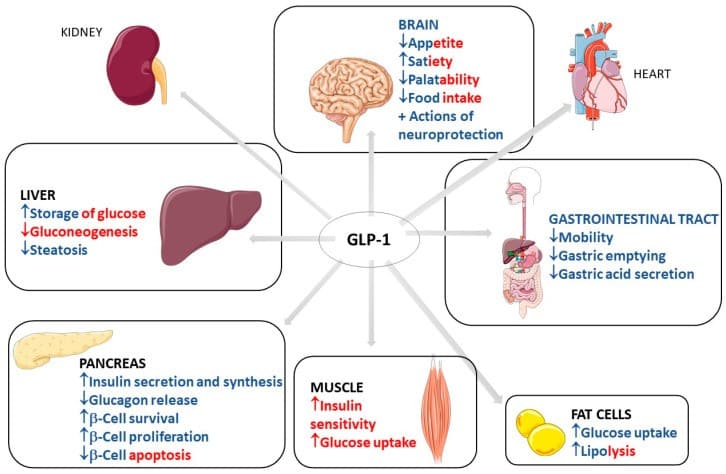
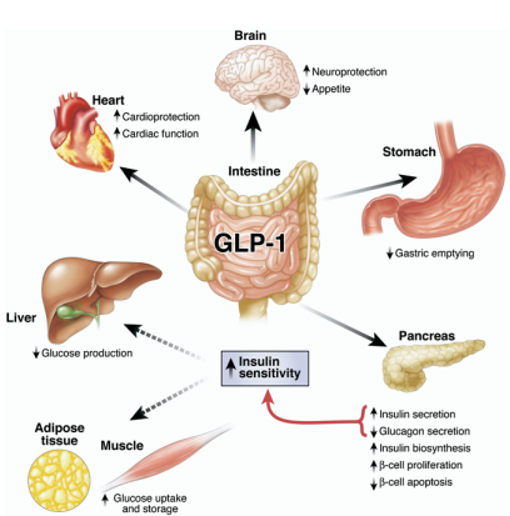
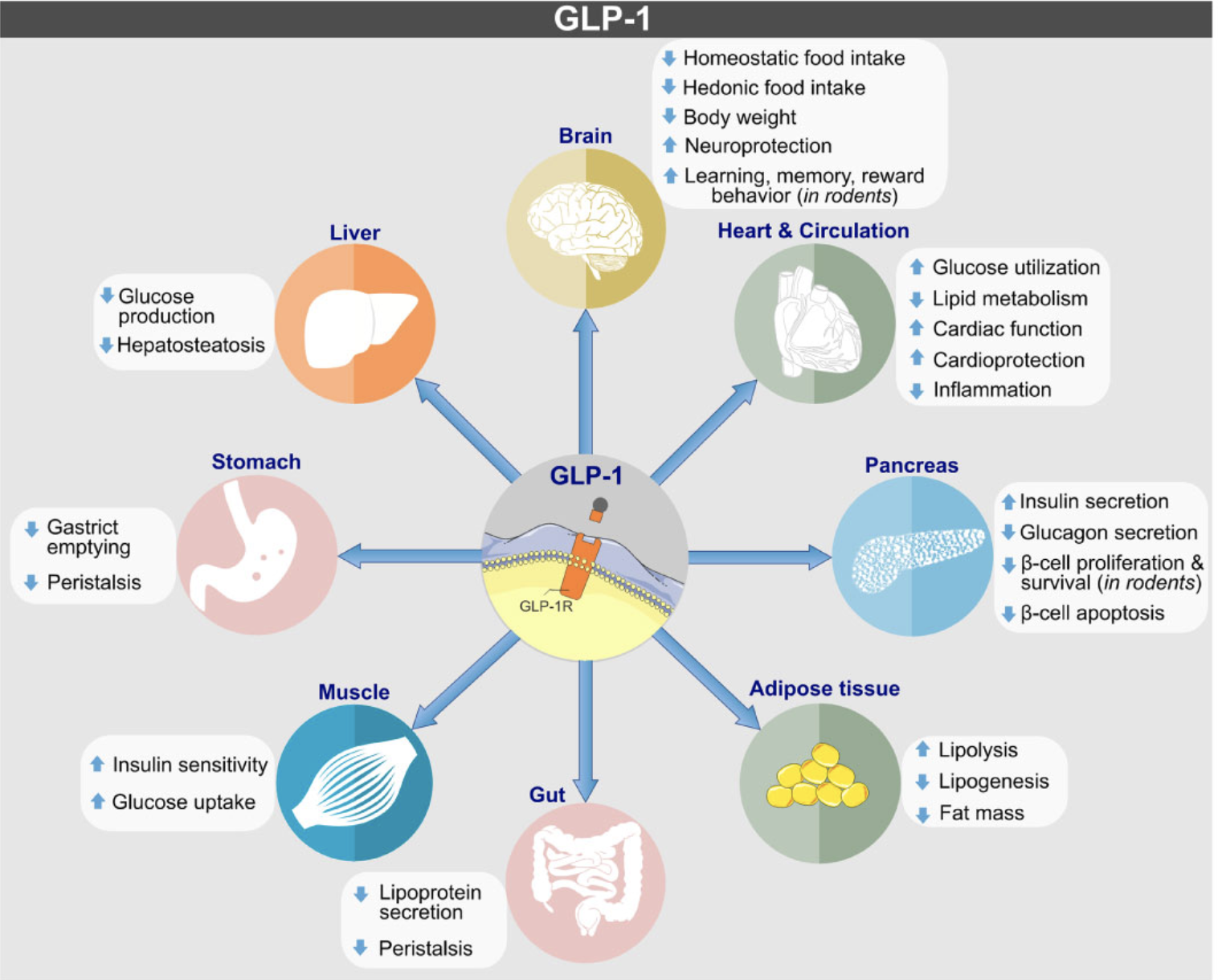
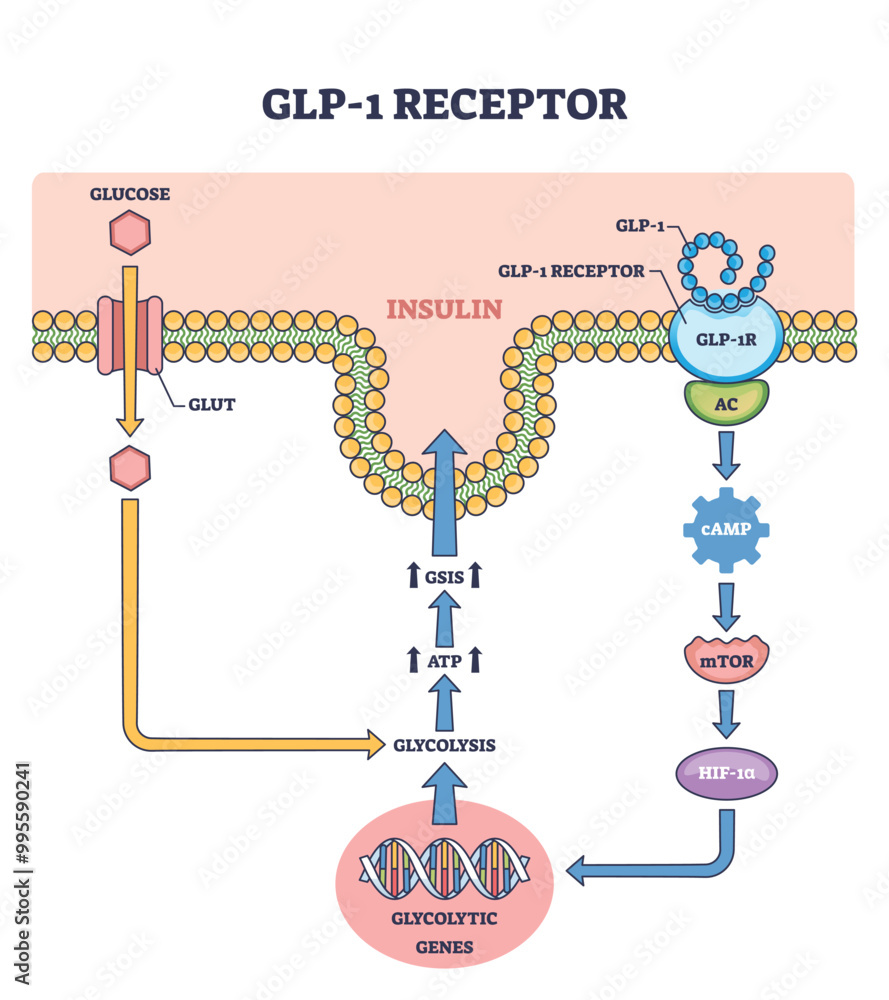
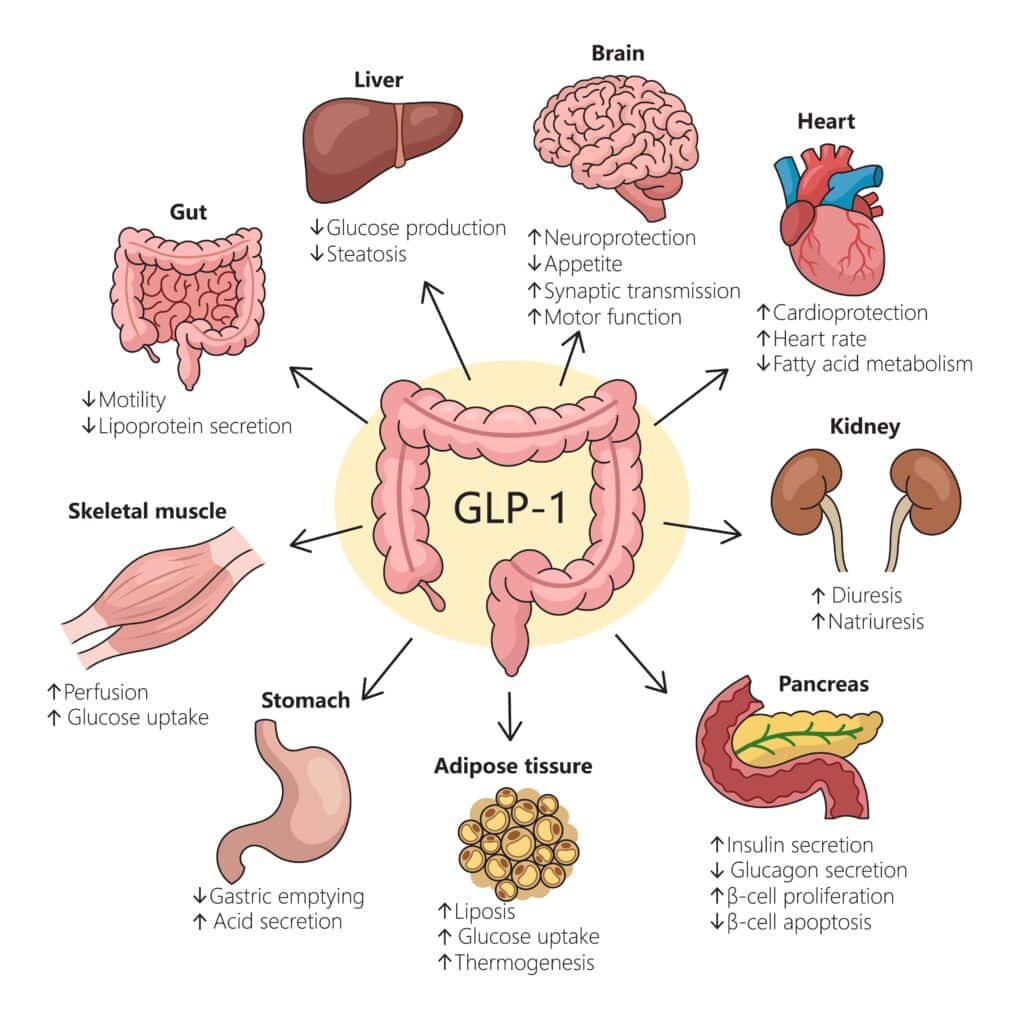
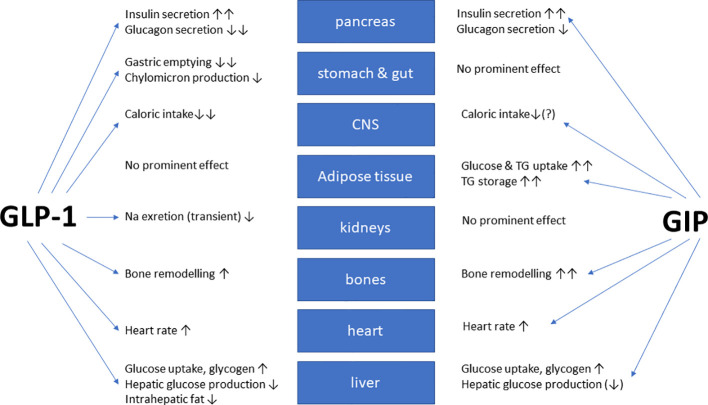
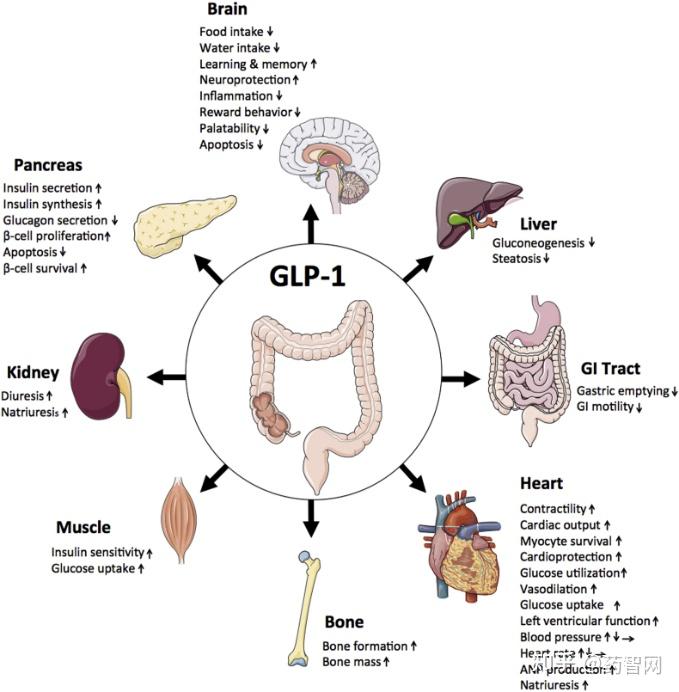
GLP-1 is a gastrointestinal peptide that plays a central role in glucose metabolism. It is secreted by L cells in the intestine in response to food intake, and its postprandial secretion is triggered by nutrient-sensing via transporters and G-protein-coupled receptors (GPCRs). GLP-1 has been shown to enhance insulin secretion, suppress glucagon release, and promote weight loss, making it an attractive therapeutic target for diabetes management.
The Impact of GLP-1 on Fastings in Kids with T1D
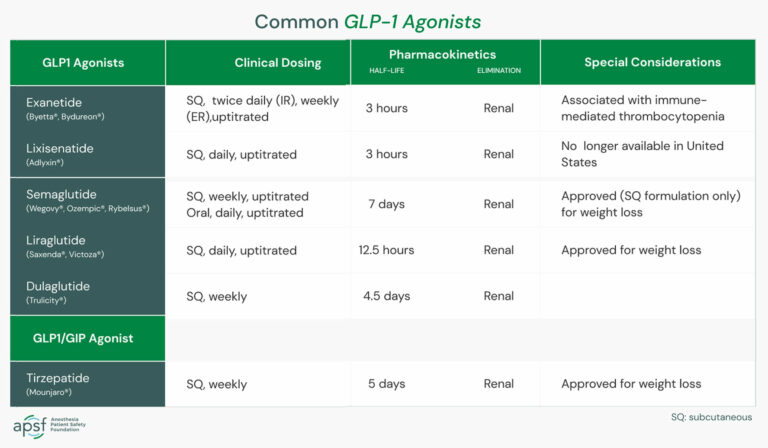
Research on the impact of GLP-1 on fasting in kids with T1D is limited, but some studies suggest that GLP-1 levels may be lower in children with T1D compared to healthy controls. A study published in the Journal of Clinical Endocrinology and Metabolism found that GLP-1 levels were lower in children with T1D compared to healthy controls, and that GLP-1 levels were negatively correlated with disease duration and glycemic outcomes.
Benefits of GLP-1 Agonists in T1D Management
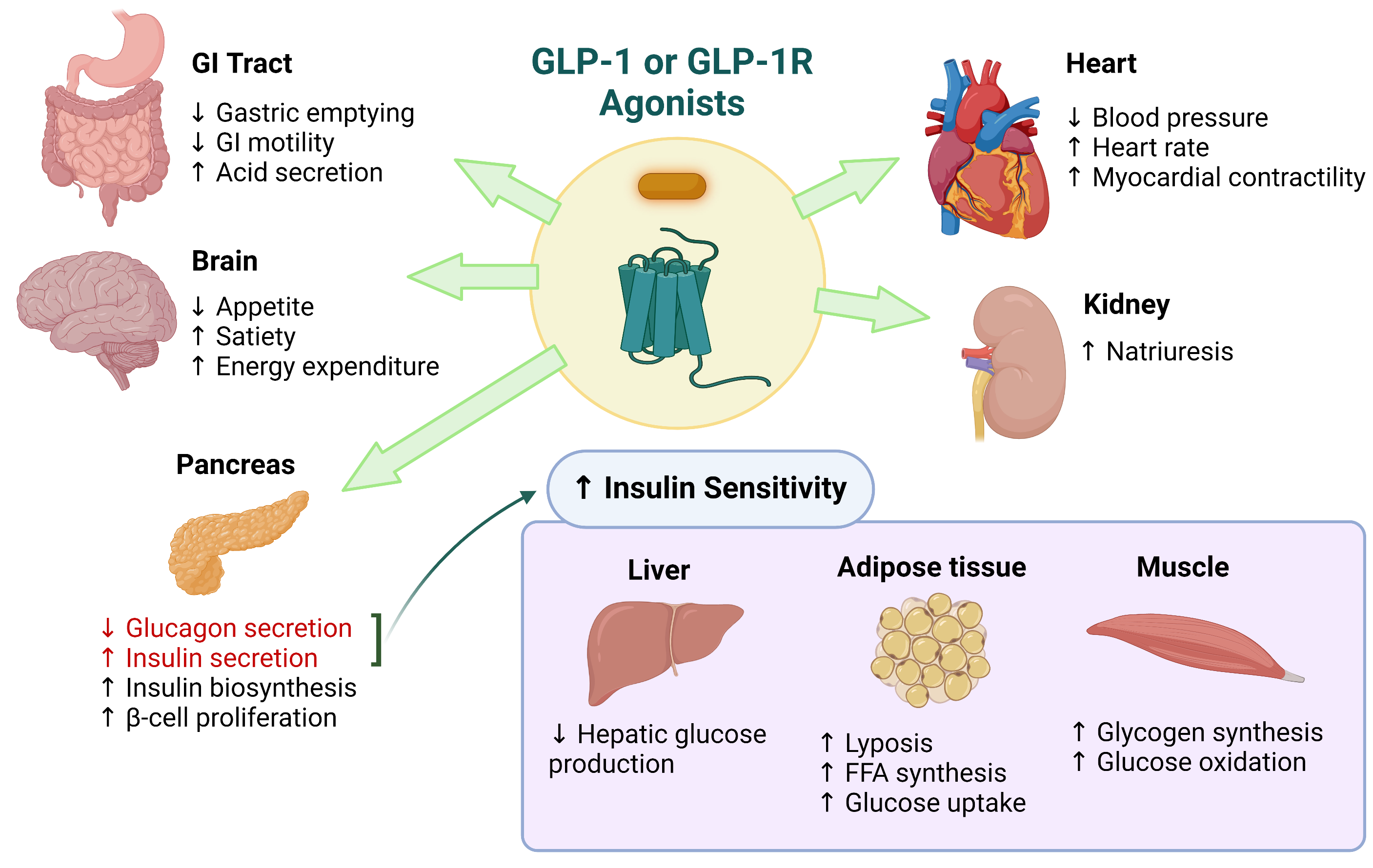
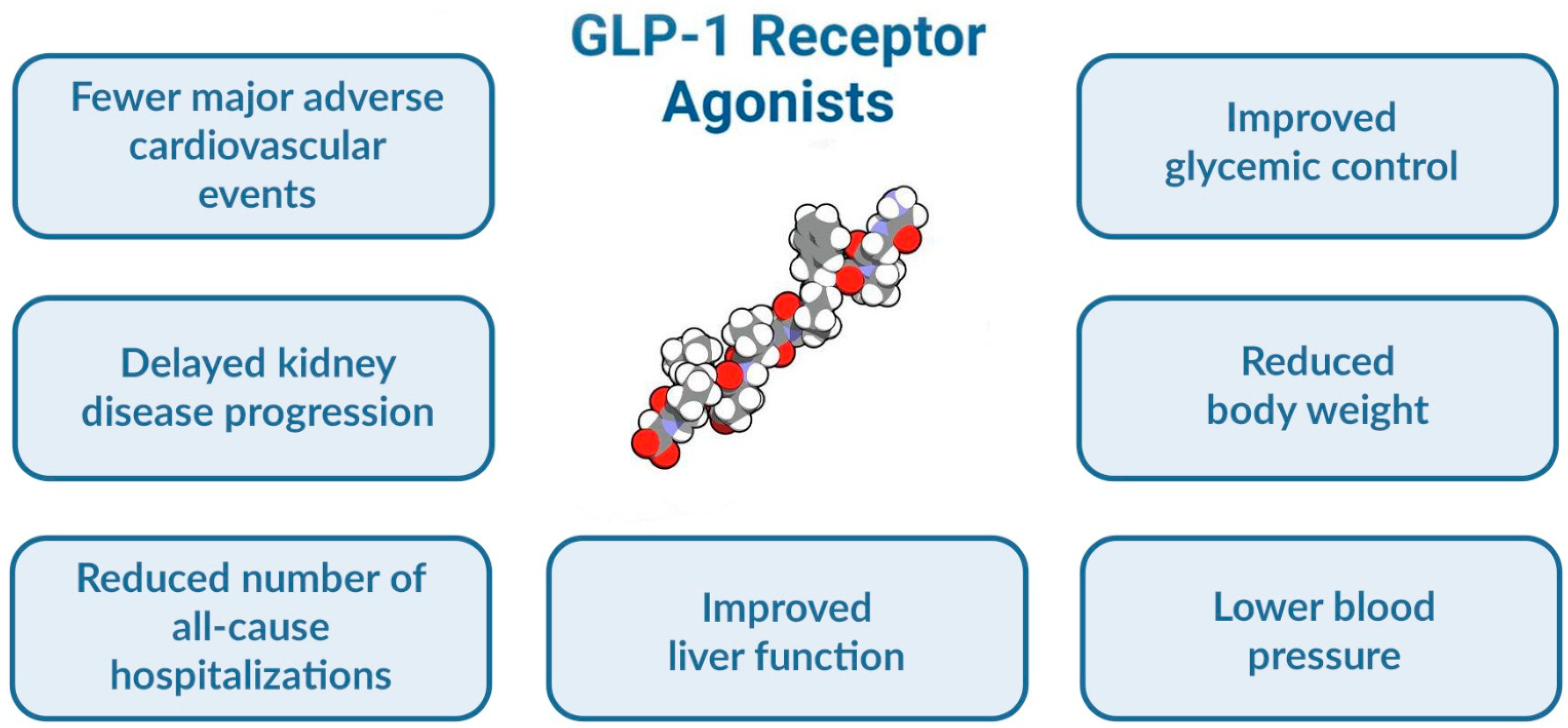
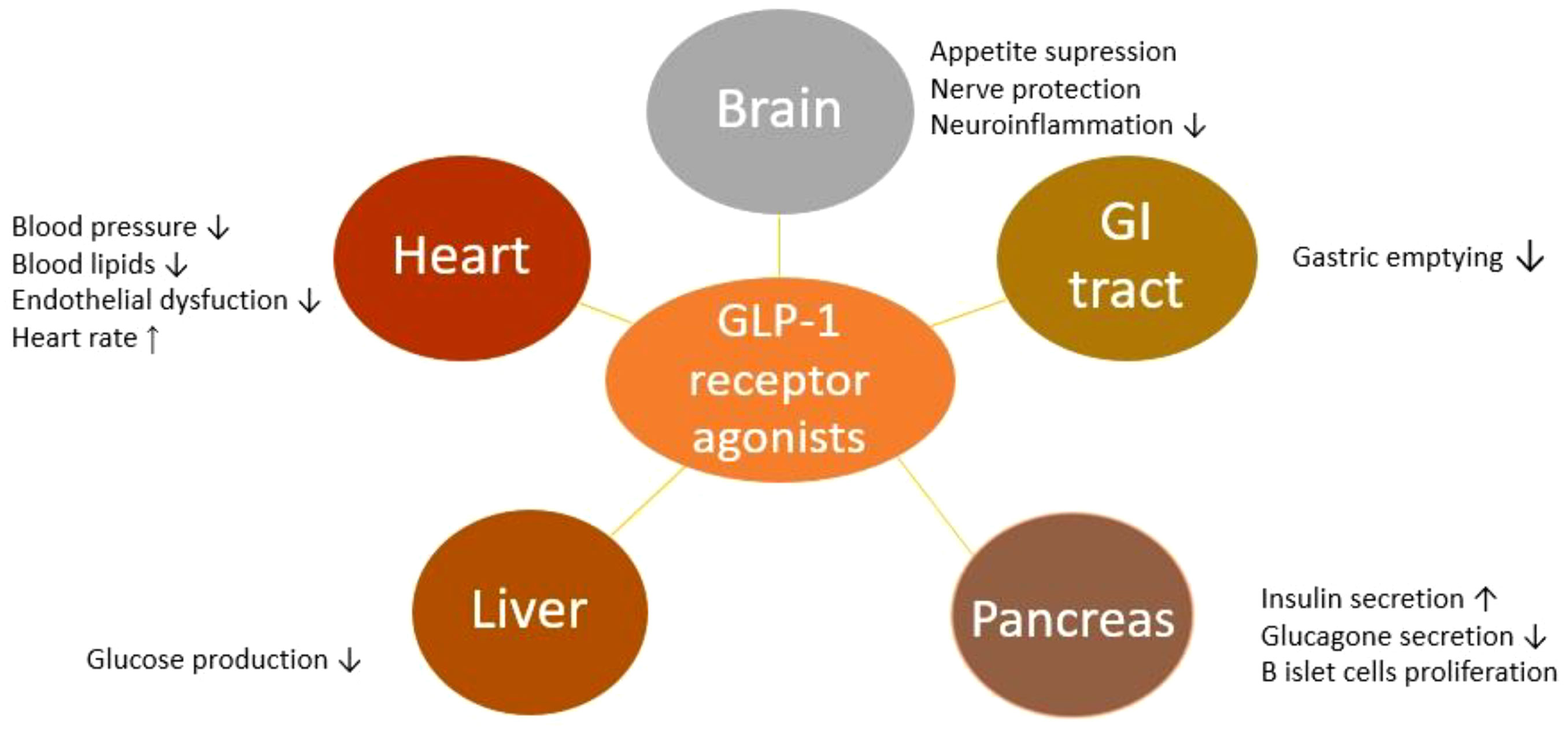
- Improved glycemic control: GLP-1 agonists have been shown to lower HbA1c levels and reduce the need for insulin in adults with T1D.
- Weight loss: GLP-1 agonists have been shown to promote weight loss in children and adolescents with obesity, which can improve glycemic control and reduce the risk of complications.
- Cardiorenal protection: GLP-1 agonists have been shown to have cardiorenal protective effects in adults with T1D, reducing the risk of cardiovascular disease and kidney damage.
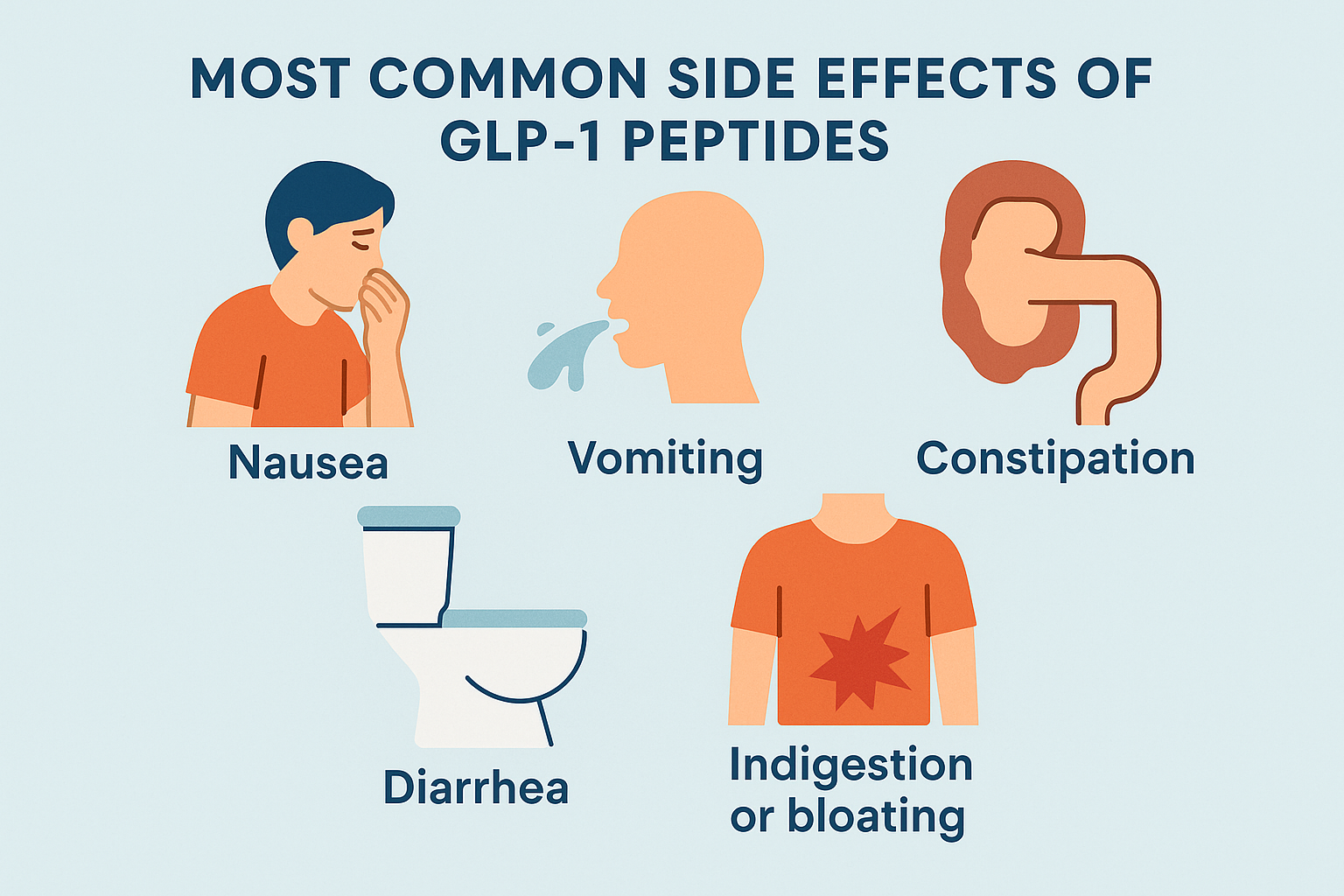
While the current evidence on GLP-1 agonists in T1D management is limited, ongoing studies are investigating the efficacy and safety of GLP-1 agonists in children with T1D. A Phase 3 clinical trial is currently underway to evaluate the efficacy and safety of maridebart cafraglutide, a long-acting peptide-antibody conjugate designed to increase GLP-1 receptor activity while inhibiting GIP receptor activity, in children and adolescents with T1D.

Conclusion
References:
1 et al. Comparison of incertin levels in children and adolescents with T1D and healthy controls: a systematic review. Journal of Clinical Endocrinology and Metabolism. 2022.
2 et al. Glucagon-like peptide-1 receptor agonists in the treatment of type 2 diabetes mellitus. European Journal of Endocrinology. 2021.
3 et al. Effects of glucagon-like peptide-1 receptor agonists on obesity and cardiorenal protection in children and adolescents with T1D. Pediatrics. 2022.