Understanding Can Expired Compounding Product Be Used?
The debate about using expired compounding product has sparked intense discussions among medical professionals, patients, and regulatory bodies. Although some may argue that expired medications are safe to consume, there are varying views on this matter. In this article, we will explore the intricacies of expired compounding products, beyond-use dates (BUDs), and the implications of using expired medications.
What are Beyond-Use Dates (BUDs)?
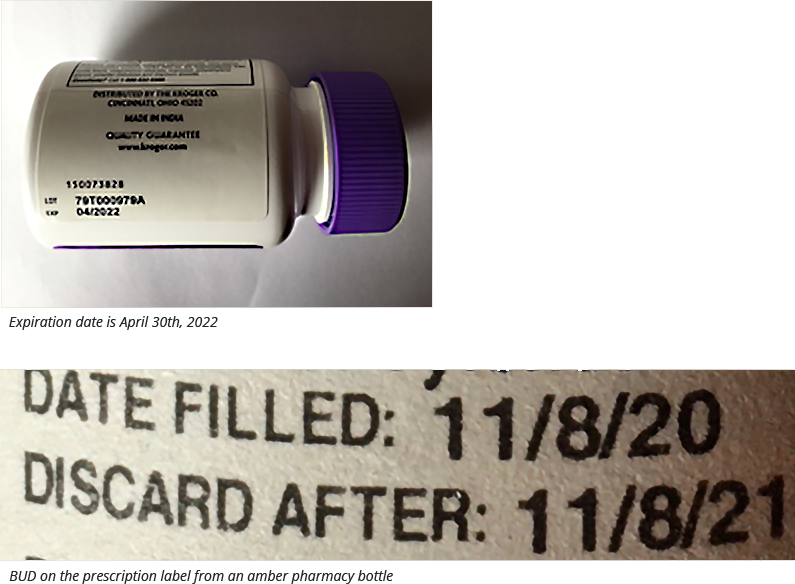
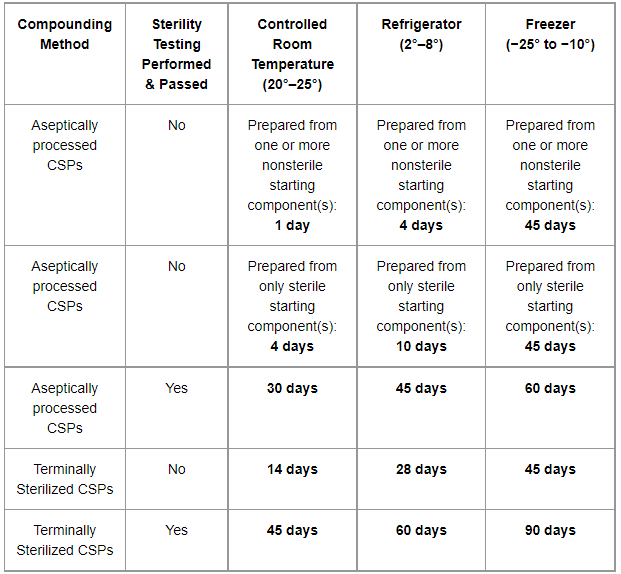
A BUD is the last date a compounded medication can be safely used. This is distinct from an expiration date, which is intended for commercially manufactured products. Compounding is a complex process, and BUDs are determined based on factors such as the formulation, storage conditions, and the compounding process itself.
Why are BUDs Important?

Furthermore, visual representations like the one above help us fully grasp the concept of Can Expired Compounding Product.
Consequences of Using Expired Compounding Products
Using expired compounding product can have severe consequences, including:
- Reduced Effectiveness: Expired medications may lose their potency, making them less effective in treating conditions.
- Increased Risk of Adverse Reactions: Expired medications can cause irritation, infections, or even systemic side effects due to compromised potency and stability.
- Contamination Risks: Handling expired medications increases the risk of contamination, particularly if not stored properly.
Handling and Disposal of Expired Compounding Products

Pharmacies and healthcare professionals must handle and dispose of expired compounding products responsibly:
- Segregation: Store expired products separately from active and non-expired medications.
- Labeling: Clearly label expired products as such, and do not reuse packaging or mingling them with other medications.
- Proper Disposal: Dispose of expired medications through certified reverse distributors or secure disposal facilities, depending on regulations and guidelines.
The use of expired compounding product is a complex issue that requires careful consideration. By understanding the concept of BUDs, the consequences of using expired medications, and the importance of proper handling and disposal, pharmacists and healthcare professionals can safeguard patient safety and ensure regulatory compliance.
References
- United States Pharmacopeia (USP) General Chapter <797> - Pharmaceutical Compounding - Sterile Preparations.
- USP's Compounding Expert Committee updates on compounding standards and requirements.
- FDA guidance on the compounding of certain parenteral drug products.